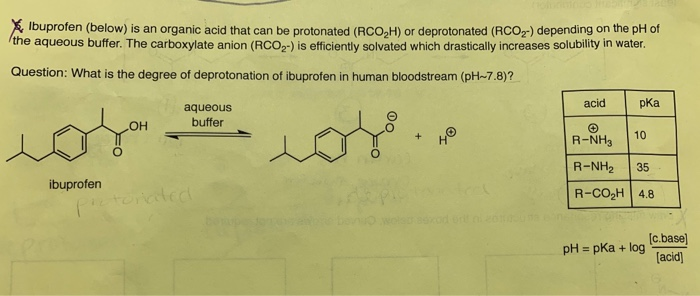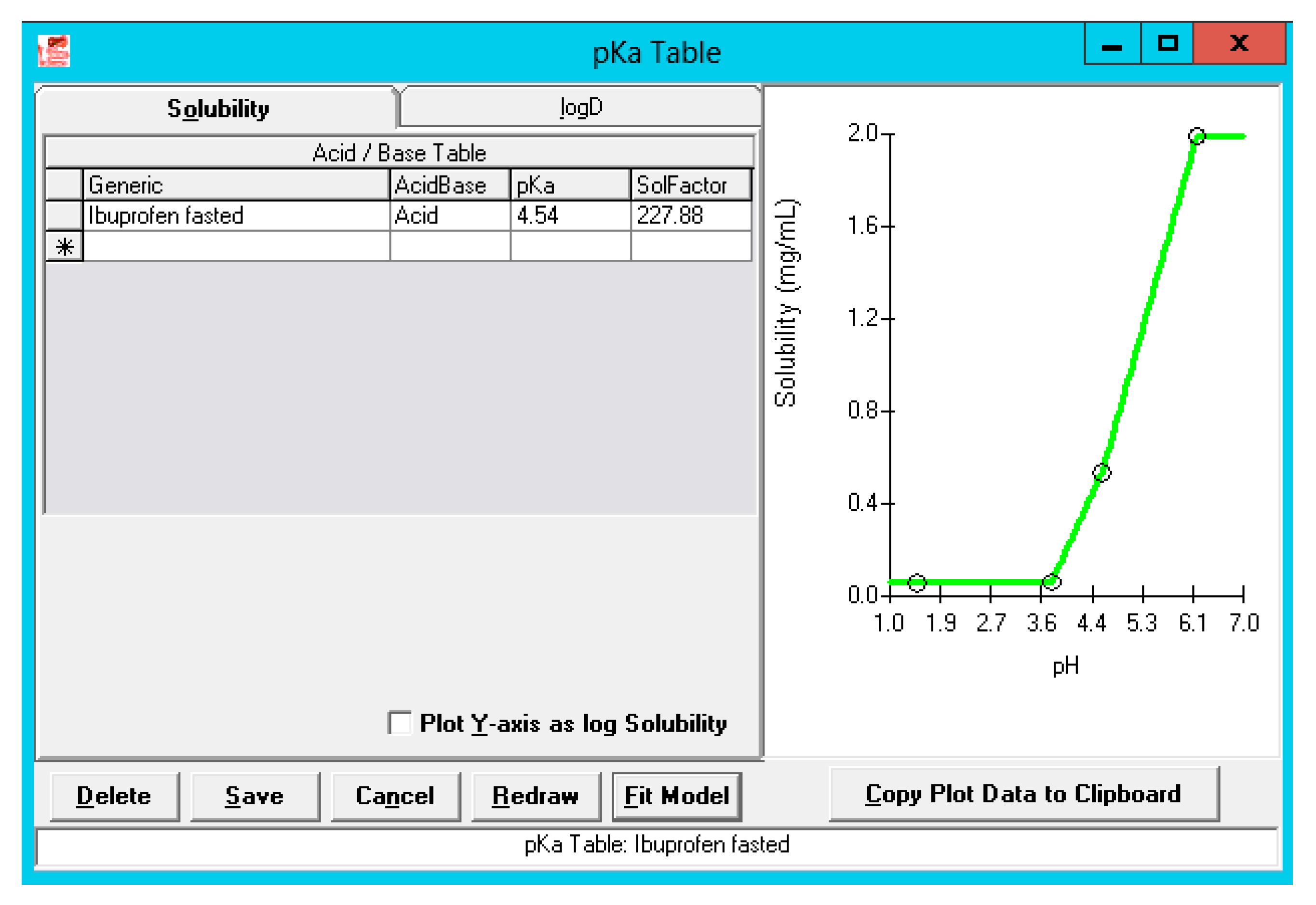
Pharmaceutics | Free Full-Text | A Mechanistic Physiologically-Based Biopharmaceutics Modeling (PBBM) Approach to Assess the In Vivo Performance of an Orally Administered Drug Product: From IVIVC to IVIVP
![PDF] Sorption, photodegradation, and chemical transformation of naproxen and ibuprofen in soils and water. | Semantic Scholar PDF] Sorption, photodegradation, and chemical transformation of naproxen and ibuprofen in soils and water. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bd825006414bdf48d3e25685d998bf571ff563c7/2-Figure1-1.png)
PDF] Sorption, photodegradation, and chemical transformation of naproxen and ibuprofen in soils and water. | Semantic Scholar

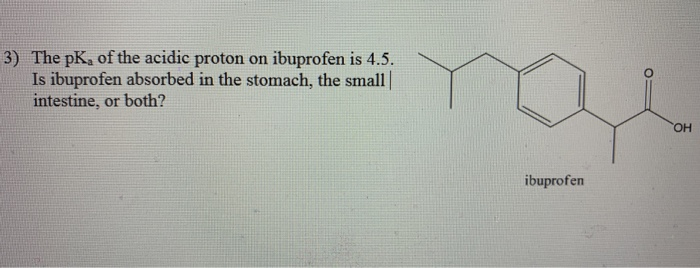
Ibuprofen (aka ADVIL) is a weak acid with a pKa of 4.9. It is absorbed through the stomach and the small - brainly.com

Effect of Water pH on the Uptake of Acidic (Ibuprofen) and Basic (Propranolol) Drugs in a Fish Gill Cell Culture Model | Environmental Science & Technology

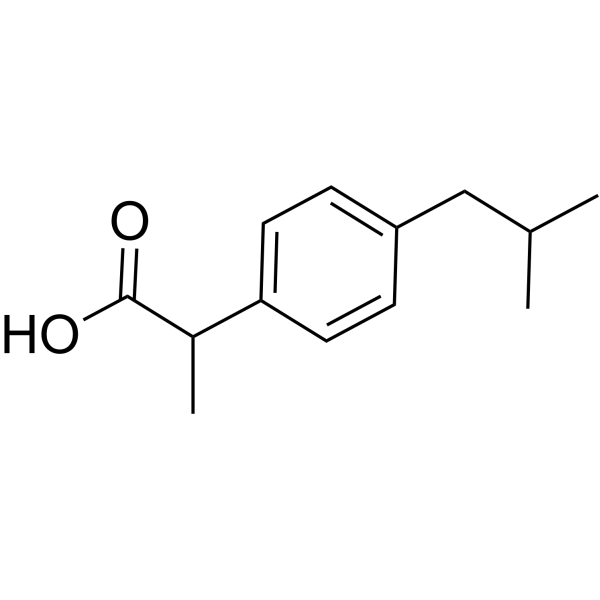
Chemical structure of ibuprofen and fenoprofen ; (*) denotes the chiral... | Download Scientific Diagram

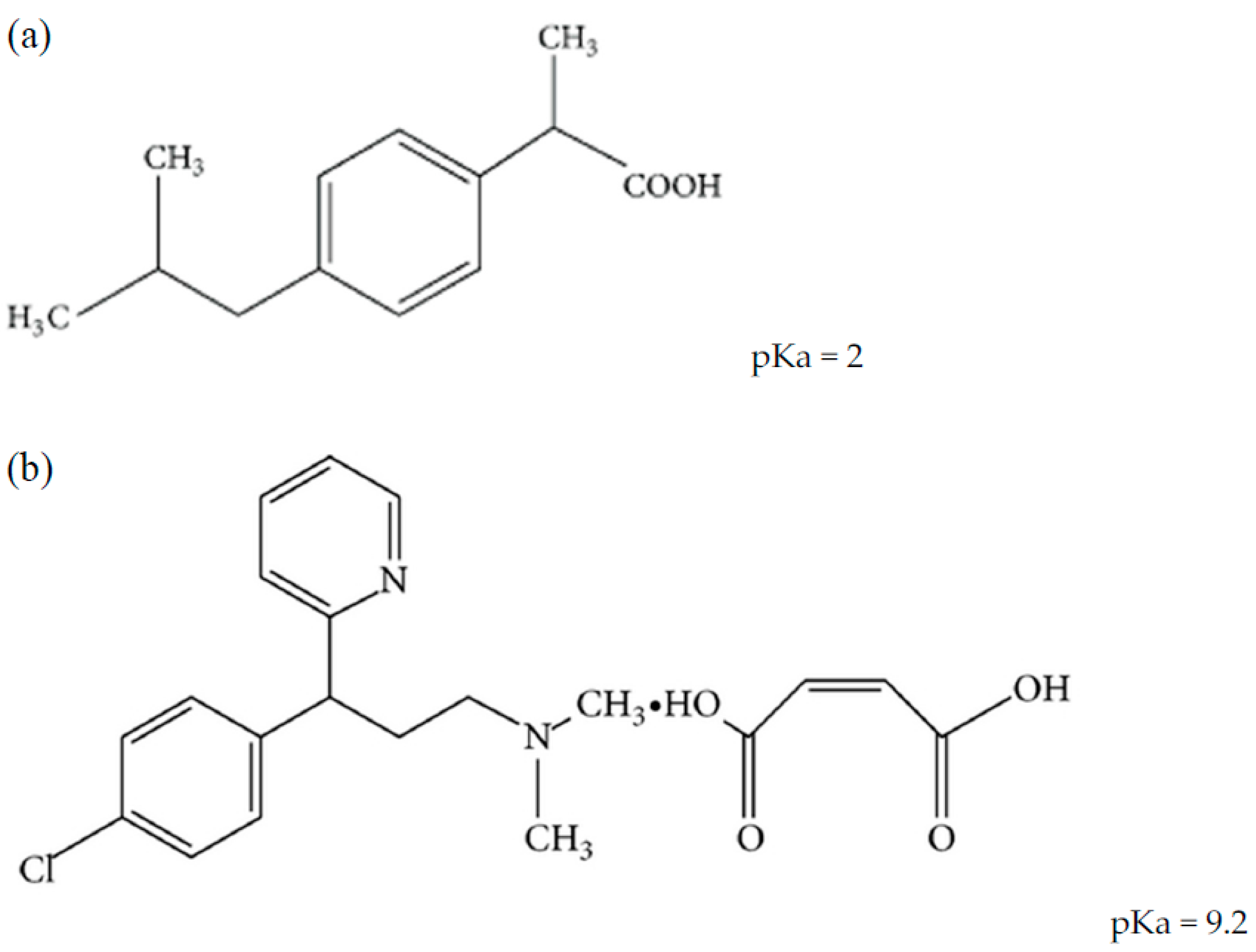
Chemical structures and pKa values of the five different nonsteroidal... | Download Scientific Diagram

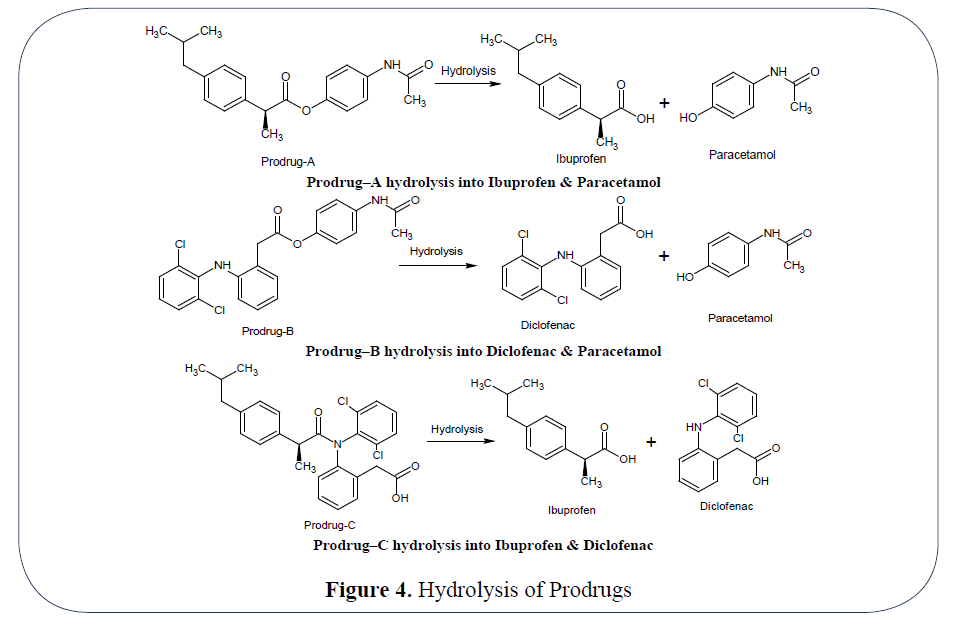
Comparative evaluation of ibuprofen co-crystals prepared by solvent evaporation and hot melt extrusion technology - ScienceDirect
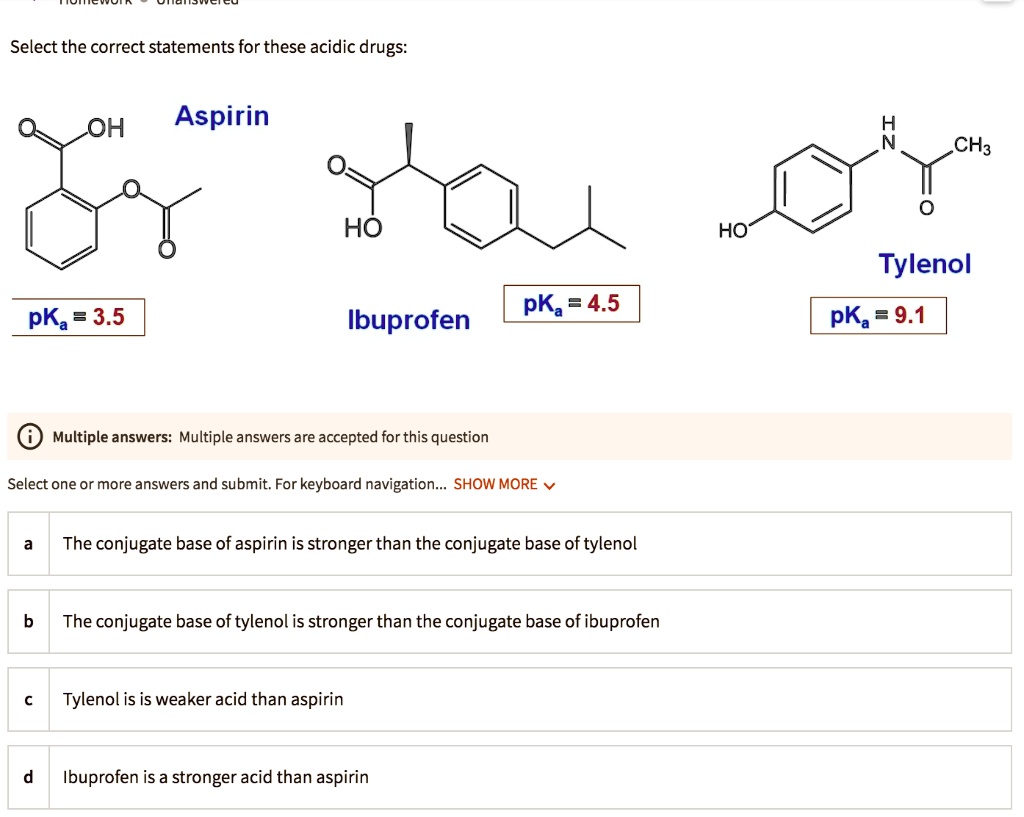
SOLVED: Select the correct statements for these acidic drugs: OH Aspirin HO HO Tylenol pKa 9.1 pKa =4.5 pKa 3.5 Ibuprofen Multiple answers: Multiple answers are accepted for this question Select one
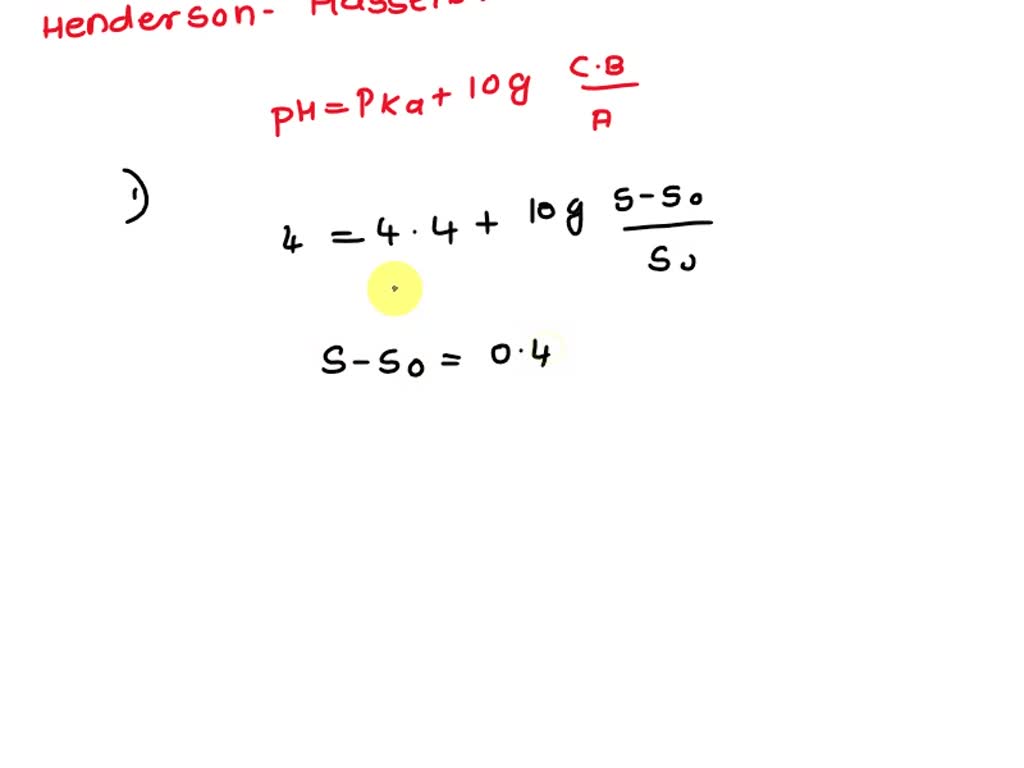
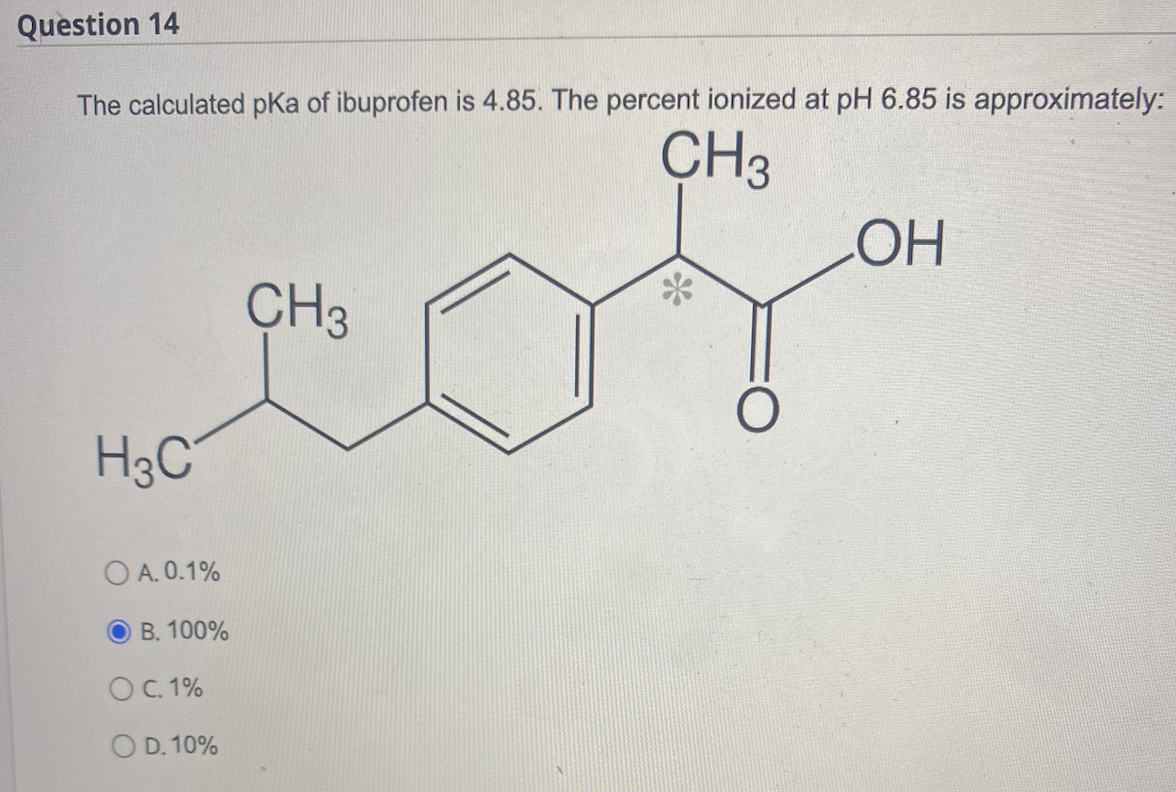
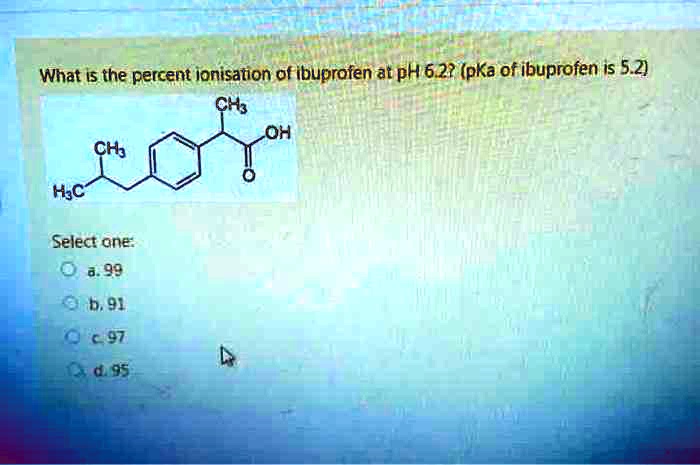
SOLVED: Ibuprofen is an acidic drug with a pKa of 4.4. The solubility of ibuprofen in water at pH 0.5 and 25 C is 5.52 x 10-5 M. At the same temperature,
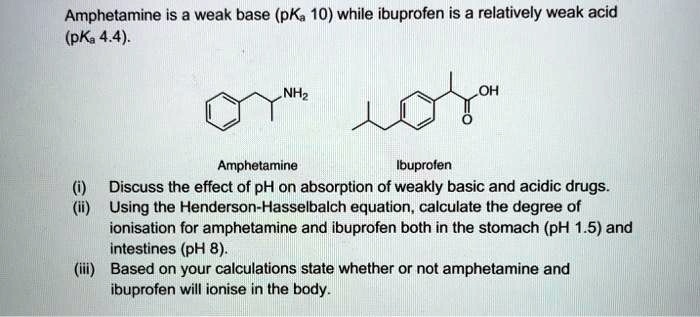
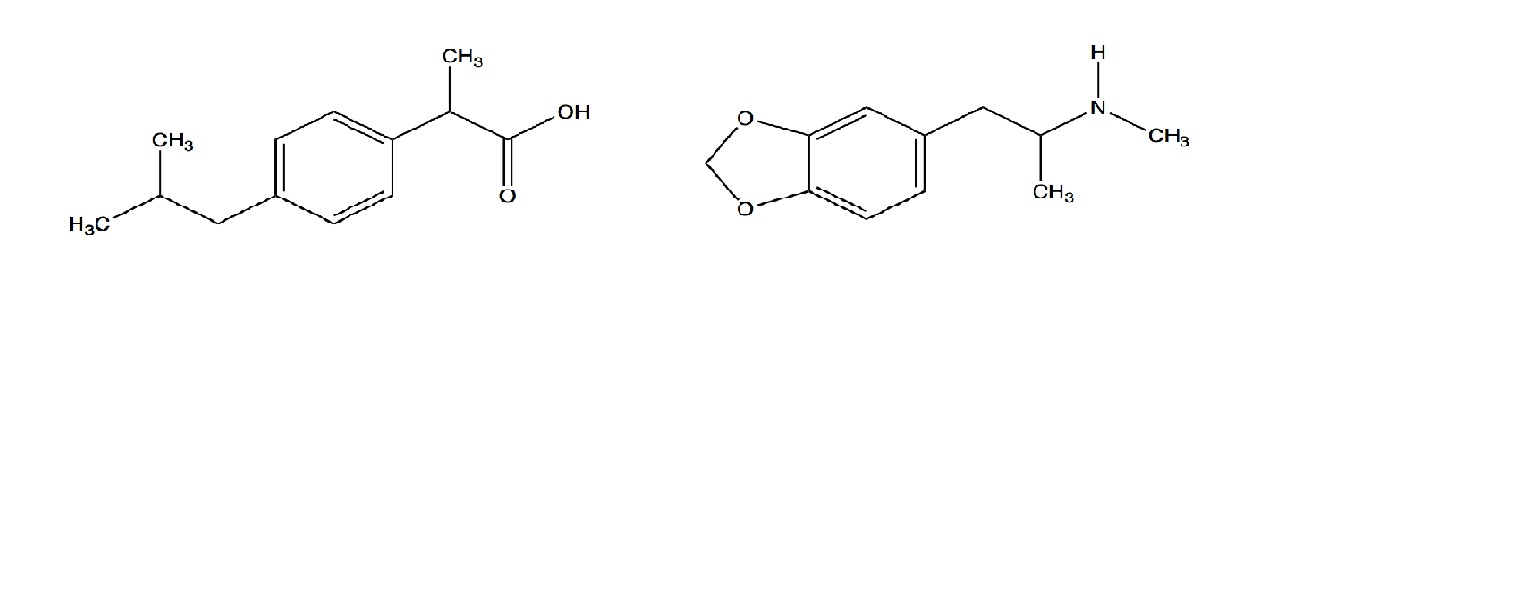
SOLVED: Amphetamine is a weak base (pKa 10) while ibuprofen is a relatively weak acid (pKa 4.4). Discuss the effect of pH on absorption of weakly basic and acidic drugs. Using the